eIFU – The Simplified and Cost-Effective Solution for Managing Digital Instructions for Use (IFU)
Simplify and accelerate the management of Instructions for Use (IFU) for your Medical Devices (MD) and In Vitro Diagnostics (IVD) with our streamlined solution.
Trust an experienced partner: Since 2021, RealBit is a member of BioPmed, the leading innovation and research hub for the medical sector in the Piedmont region.
Discover the benefits eIFU can bring to your business!
Download the Brochure Request a Demo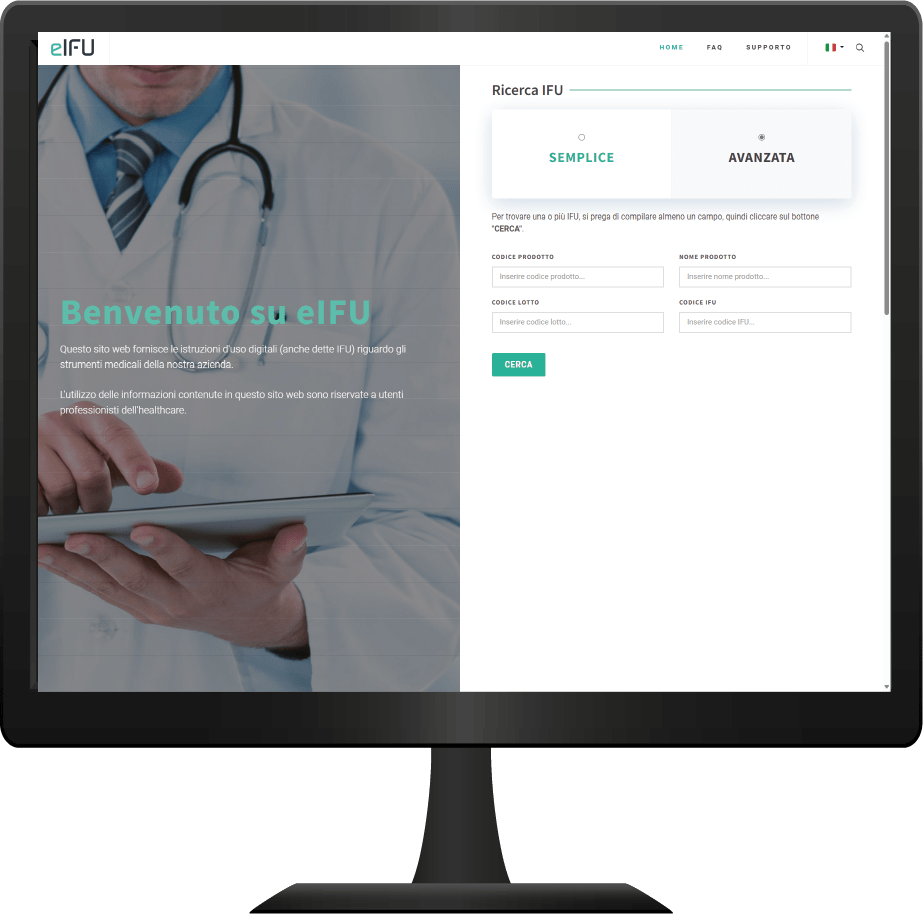
A simplified and effective solution for your instructions for use
For years, we have been working on enterprise digitalization projects, and since 2021, we have extended our expertise to the medical sector with eIFU, our software solution for managing electronic Instructions for Use (IFU).
With just a few clicks, your updated IFU revisions will be available online for your customers, ensuring full compliance with current regulations.
The Benefits of Switching to eIFU
Cost savings, optimization and sustainability
Transitioning from paper to digital with our eIFU solution brings tangible and concrete advantages in terms of cost reduction, time efficiency, and logistics optimization, all while making a meaningful contribution to environmental sustainability.
Instant Publication
Make IFU revisions available worldwide with a single click, eliminating waiting times needed for printing and shipping.
Real-Time Revision Notifications
Specialists can be automatically notified whenever a new revision is released.
Lower Material Costs
With the QR Code generated by eIFU, you can reduce packaging size and costs, increasing overall savings.
Simplified Management
Manage all your IFUs within a single online platform, accessible anytime from any device.
Regulation-Compliant and Certification-Ready
eIFU is fully compliant with industry regulations. Along with the software, we provide the necessary validation documents for certification.
Customizable Design
Tailor the interface with your brand colors and logos to strengthen your brand identity with your customers.
Trust a Specialized Partner
MeDeLiA is coming – The medical suite by RealBit
RealBit offers a complete and modular solution for companies in the medical sector.
Starting from January 2025, Post-Market Surveillance (PMS) and Post-Market Clinical Follow-up (PMCF) will become essential tools for medical device manufacturers to maintain compliance during certification. eIFU, our solution for managing electronic Instructions for Use, is evolving into a comprehensive medical suite designed to meet the diverse needs of medical device and in vitro diagnostic manufacturers.
Discover the benefits of navigating digital transformation and adapting to new regulations with a single, trusted technology partner.
Since 2021, RealBit is a proud member of BioPmed, the leading innovation cluster for the medical sector.
Key Features
Discover the main strengths of our software
Here are the key features our clients appreciate the most:
IFU access via QR Code – Scan to view or download instantly.
Simplified IFU Management – A centralized platform for seamless handling.
Multi-language, products and batch management – Ensure accessibility across global markets.
Country-specific revision management – Adapt IFUs to regulatory requirements by region.
Audit process simplification – Streamline compliance and regulatory checks.
Revision tracking & management – Maintain full control over document updates.
Customizable design – Tailor the interface to reflect your brand identity.
Validation documents provided & certification-ready software – Ensuring compliance with industry standards.
Fully compliant with FDA & MDR regulations – Meeting the highest regulatory requirements.

Available in Cloud or On-Premise

Choose eIFU in the Cloud, hosted on RealBit’s medical-compliant servers, designed for high reliability and constant backups.

Install eIFU on your company’s servers—download the brochure to learn more about hardware requirements.
REALBIT TIPS
Key considerations for transitioning to eIFU
Regulatory compliance
Determine whether electronic IFUs are accepted for the type of products you manufacture and in the countries where you sell them. Anyhow, eIFU allows your customers to request a printed copy if needed, as required by regulations.
Our eIFU solution complies with EU Regulation (EU) 2017/745 and is provided with the necessary validation. Once you choose eIFU, the software is ready to use, and with the validation documents supplied by RealBit, you can confidently proceed with the certification process.
Evaluate your hardware solution
Check if your servers comply with regulations by downloading the brochure, or opt for the cloud solution. RealBit will provide a server configured according to the required standards for the medical industry.
Manage translations and customize the design
With eIFU, you can provide your customers with IFUs directly in their language or offer a single multilingual file. Customize the design of the client platform with colors, images, and videos to maintain your brand identity.
The Future of IFUs
An evolving regulatory landscape
The digitalization of Instructions for Use (IFU) is becoming increasingly prevalent in the medical device (MD) and in vitro diagnostic (IVD) manufacturing across Europe and worldwide. Regulatory authorities are focusing on monitoring the process to ensure benefits for the entire supply chain, from manufacturers to end users—while improving quality and sustainability compared to paper versions.
RealBit can support this transition by providing a software solution that is already validated according to the latest regulations (FDA & MDR) and certification-ready.
In this section, you will find the regulations and guidelines that eIFU complies with.
Regulation (EU) 2017/745 of April 5, 2017, on medical devices, amending Directive 2001/83/EC, Regulation (EC) No. 178/2002, and Regulation (EC) No. 1223/2009, and repealing Council Directives 90/385/EEC and 93/42/EEC.
Regulation (EU) 2017/746 of April 5, 2017, on in vitro diagnostic medical devices, repealing Directive 98/79/EC and Commission Decision 2010/227/EU.
MEDDEV 2.14/3 rev.1 – Guidelines for the provision of Instructions for Use (IFU) and other information for in vitro diagnostic medical devices (IVD).
Commission Implementing Regulation (EU) 2021/2226 of December 14, 2021, establishing rules for the implementation of Regulation (EU) 2017/745 regarding the electronic Instructions for Use (IFU) of medical devices.
Blog
Leggi i nostri ultimi articoli
Smart software for your business!
Contact us today – we’ll guide you through each implementation phase with full support. Learn more by downloading our brochure or request a demo presentation!






